BONLAB BLOG
Thoughts
&
Scientific Fiction
BonLab fabricates textured microcapsules through crystallization
Watch the Talk by clicking here.
Microcapsules can be found in a range of commercial applications, including cosmetics, healthcare, agriculture, and food. The capsules serve as a storage vessel for an active ingredient, for example a nutrient or fragrance. They can have a variety of designs, the simplest form being a single internal liquid-based core surrounded by a solid shell. The chemical and physical characteristics of this shell influence the colloidal stability of capsules in formulations, dictate the permeability and mechanical robustness of the capsules, and can potentially regulate substrate adhesion. Beside storage, these features of the microcapsules are there to regulate and control release and delivery of the active compound.
A considerable part of the technologies used to produce microcapsule relies on the use of synthetic polymers that do not break down, with terrible consequences for environmental build up. One way is to make use of biocompatible and degradable plastics.
We provide an alternative solution, in that we fabricate the capsule from small molecular compounds, instead of polymers, that can crystallize.
Microcapsules can be found in a range of commercial applications, including cosmetics, healthcare, agriculture, and food. The capsules serve as a storage vessel for an active ingredient, for example a nutrient or fragrance. They can have a variety of designs, the simplest form being a single internal liquid-based core surrounded by a solid shell. The chemical and physical characteristics of this shell influence the colloidal stability of capsules in formulations, dictate the permeability and mechanical robustness of the capsules, and can potentially regulate substrate adhesion. Beside storage, these features of the microcapsules are there to regulate and control release and delivery of the active compound.
A considerable part of the technologies used to produce microcapsule relies on the use of synthetic polymers that do not break down, with terrible consequences for environmental build up. One way is to make use of biocompatible and degradable plastics.
We provide an alternative solution, in that we fabricate the capsule from small molecular compounds, instead of polymers, that can crystallize.
Artist impression of textured microcapsules made through crystallization.
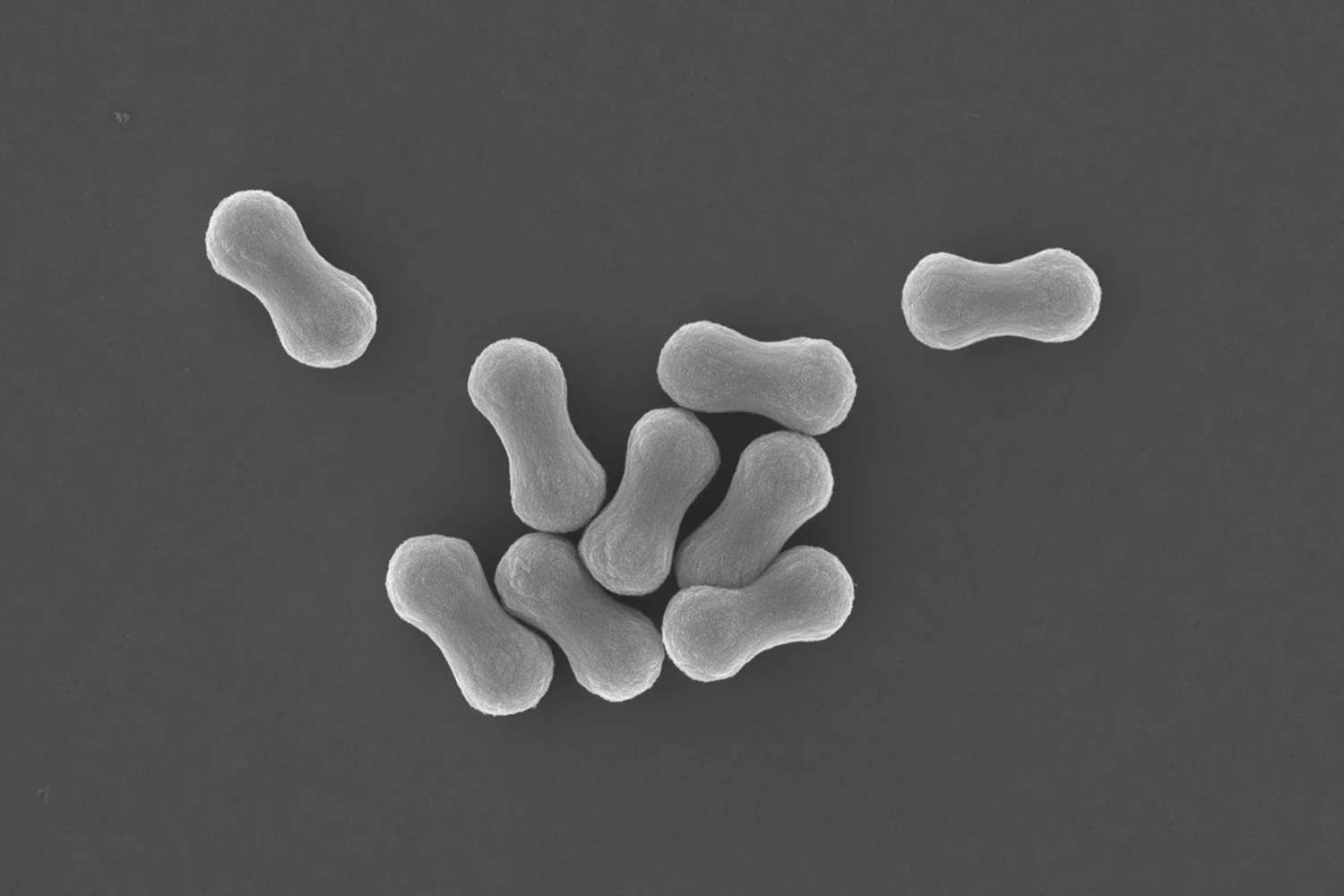
In our work, published in the scientific journal ACS Applied Materials & Interfaces we focus on the fabrication of microcapsules of which the capsule wall is composed of an inter-locked mesh of needle-like crystals, which besides capsule rigidity and semi-permeability, provides a roughened surface texture.
In this proof-of-concept study crystals of the small organic compound decane-1,10-bis(cyclohexyl carbamate) are formed within the geometric confinement of emulsion droplets, through precipitation from a binary-solvent dispersed phase. This binary mixture consists of a volatile solvent and non-volatile carrier oil. Crystallization is facilitated upon supersaturation due to evaporation of the volatile solvent. We show that the microcapsule diameter can be easily tuned using microfluidics.
The technology is shown to be scalable using conventional mixers, yielding spikey microcapsules with diameters in the range of 10-50 μm. We highlight that the capsule shape can be molded and arrested by jamming using recrystallization in geometric confinement.
With an eye on commercial applications we show that these textured microcapsules show enhanced deposition onto a range of fabric fibers. They outperform their traditional polymeric analogues by sticking better on fibrous substrates.
Prof. dr. ir. Stefan Bon says: “I am delighted with this work, a coordinated effort which was led by first author dr. Sam Wilson-Whitford. A while back we decided in BonLab that our science needed to be greener, and more sustainable. We think our way of making textured microcapsules meets these criteria. We hope that it inspires industry, and that the concept will prove to be useful across a variety of application areas.”
The paper can be accessed here:
https://pubs.acs.org/doi/10.1021/acsami.0c22378
A talk on the topic can be accessed here:
https://www.formulation.org.uk/pc20programme/250-past/2021/pc20/860-pc20-bon.html
New method to study chain transfer in radical polymerizations
Synthetic polymers in most cases do not have one bespoke molecular weight. A sample typically consists of a large number of individual polymer chains, each having a different molecular weight. The average molecular weights and the shape of the molecular weight distribution are a kinetic fingerprint of how to polymer material was made. The resulting molecular weight distribution dictates physical and mechanical properties.
In free radical polymerizations, four key mechanistic events need to be considered. These are initiation, propagation, termination, and chain transfer. The latter often gets brushed under the carpet in introductory textbooks, but is pivotal.
When one targets polymers of low molecular weight, chain transfer agents are often used. One prominent class of chain transfer agents are thiol compounds, for example n-dodecanethiol. To understand how the molecular weight distribution develops throughout the polymerization process, the ability to determine the reactivity of the chain transfer agent is crucial. This reactivity is often expressed in the form of a chain transfer constant, Ctr, which is the ratio of the rate coefficients of chain transfer and propagation.
Synthetic polymers in most cases do not have one bespoke molecular weight. A sample typically consists of a large number of individual polymer chains, each having a different molecular weight. The average molecular weights and the shape of the molecular weight distribution are a kinetic fingerprint of how to polymer material was made. The resulting molecular weight distribution dictates physical and mechanical properties.
In free radical polymerizations, four key mechanistic events need to be considered. These are initiation, propagation, termination, and chain transfer. The latter often gets brushed under the carpet in introductory textbooks, but is pivotal.
When one targets polymers of low molecular weight, chain transfer agents are often used. One prominent class of chain transfer agents are thiol compounds, for example n-dodecanethiol. To understand how the molecular weight distribution develops throughout the polymerization process, the ability to determine the reactivity of the chain transfer agent is crucial. This reactivity is often expressed in the form of a chain transfer constant, Ctr, which is the ratio of the rate coefficients of chain transfer and propagation.
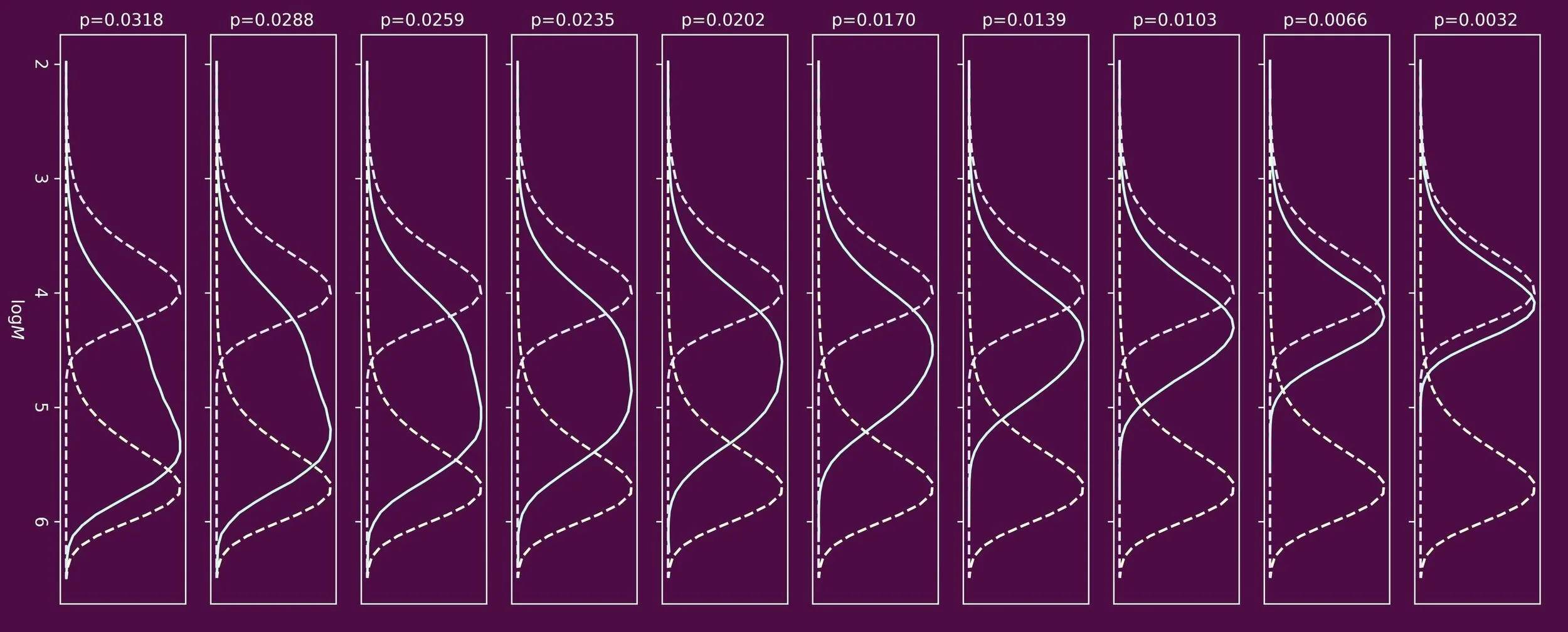
The most famous method to determine values for the chain transfer constant, Ctr, is referred to as the Mayo method. It is an excellent method, but over time its assumptions and boundary conditions have been eroded. The Mayo method is only valid if there is no marked composition drift, that is a drift in concentration ratio of chain transfer agent and monomer. In essence it makes use of the instantaneous molecular weight distribution, here in the form of the number average molecular weight. Whereas this is easy to achieve in experiments where Ctr < 1 by keeping monomer conversion low, this is less easy to do in cases where Ctr > 1.
In the same time period (1940s) Smith already reported a solution. Whereas elegant, in this method there is no direct link with the molecular weight distribution, which may sit uncomfortably.
In our paper entitled When Mayo falls short (Ctr ≫ 1): the use of cumulative chain length distribution data in the determination of chain transfer constants (Ctr) for radical polymerizations published in the RSC journal Polymer Chemistry we propose a new method. Here we make use of the cumulative molecular weight distribution, hereby taking into account composition drift.
The validity of our method is demonstrated using Monte Carlo simulations and its experimental usefulness is shown studying one of the most challenging systems of all: the free radical polymerization of vinyl acetate in presence of thiols as chain transfer agent. We are happy to report a value for the chain transfer constant of dodecanethiol of 223, at 333 K.
Prof. dr. ir. Stefan Bon says: “ I am delighted with the final version of the manuscript. The hard work by PhD student Matt Donald to carry out the experiment and his dedication in bringing together wet science with Monte Carlo simulations paid off. We both hope that our method will be embraced, tested, and used by others. “
Details on the paper can be found here: